| Issue |
SICOT-J
Volume 12, 2026
|
|
|---|---|---|
| Article Number | 26 | |
| Number of page(s) | 6 | |
| Section | Knee | |
| DOI | https://doi.org/10.1051/sicotj/2026014 | |
| Published online | 12 May 2026 | |
Surgical Technique
Single-stage total knee arthroplasty revision with extensor mechanism allograft: surgical technique
1
Orthopaedics Surgery and Sports Medicine Department, FIFA Medical Center of Excellence, Croix-Rousse Hospital, Lyon North, University Hospital, 69004 Lyon, France
2
Univ Lyon, Claude Bernard Lyon 1 University, IFSTTAR, LBMC UMR_T9406 69622 Lyon, France
3
LIBM – EA 7424, Interuniversity Laboratory of Human Movement Science, Université Lyon 1, 69008 Lyon, France
* Corresponding author: This email address is being protected from spambots. You need JavaScript enabled to view it.
Received:
1
January
2026
Accepted:
2
March
2026
Abstract
Introduction: Chronic rupture of the extensor mechanism is a serious complication that could occur in the context of revision total knee arthroplasty (TKA). Performing combined extensor mechanism allograft reconstruction and revision TKA as a single-stage procedure requires precise surgical technique. Technique: Allograft size and quality were assessed preoperatively. A tibial tubercle osteotomy (TTO) was performed prior to revision TKA. After implantation of the definitive prosthesis, the TTO was fixed to the tibia using screws. The quadriceps tendon was sutured to the allograft using the Pulvertaft weave technique with the knee in full extension. Native patellar retinacula and prepatellar fascia were preserved to optimize graft coverage. Postoperatively, patients were immobilized in full extension for three months before starting progressive mobilization. Results: From January 2017 to April 2024, 20 patients underwent a single-stage revision TKA with total extensor mechanism allograft and were followed for a minimum of one year. Range of motion was recovered at follow-up. Five patients (25%) had failures attributed to the allograft. Conclusion: Single-stage TKA revision combined with extensor mechanism allograft, in the context of multiply operated knees, requires meticulous stepwise execution, including secure tibial fixation, precise graft tensioning, and preservation of native soft tissue coverage, to optimize outcomes in this high-risk setting.
Key words: Extensor mechanism / Allograft / Revision total knee arthroplasty / Surgical technique
© The Authors, published by EDP Sciences, 2026
 This is an Open Access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
This is an Open Access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Introduction
Chronic rupture of the extensor mechanism is a rare but serious complication following total knee arthroplasty (TKA) [1]. Surgical management options include reconstruction with various types of allografts (i.e., Achilles tendon grafts, partial or total extensor mechanism allografts [2–5]) or, as a last resort, knee arthrodesis. Among these approaches, total extensor mechanism allografts have demonstrated promising long-term outcomes [4, 6, 7]. If rupture is associated with instability, stiffness, or implant loosening, or if it follows septic complications, the reconstruction may be performed concurrently with a TKA revision (RTKA). Combining these procedures increases the complexity and duration of the operation and exposes the patient to several complications (allograft failure, stiffness, infection). Fixing the anterior tibial tuberosity in the context of tibial implant revision surgery requires a skilled surgeon. This article describes the surgical technique and clinical outcomes of total extensor mechanism allograft reconstruction performed in a single stage with RTKA.
Material and methods
This single-center retrospective study analyzed patients who underwent surgery between January 2017 and April 2024.
Indications included chronic or recurrent extensor mechanism rupture, a history of total patellectomy, a complete or ≥ 30° active extension deficit (AED), in the presence of an indication for RTKA (Table 1). Contraindications included uncontrolled infection, inadequate soft tissue coverage, acute extensor mechanism rupture without prior repair attempts or autograft reconstruction, and patients with low functional demand.
During this period, a final cohort of 20 patients met the inclusion criteria. The surgical technique was identical for every case. Follow-up was recorded, and postoperative complications were categorized into three groups: those related to RTKA, allograft, or infection. Failure was defined as an AED of ≥ 30°, revision surgery for allograft failure, revision for infection (excluding debridement, antibiotics, and implant retention [DAIR]), or revision for TKA loosening. Statistical analysis was conducted using XLSTAT software. Survival rates were estimated using the Kaplan–Meier method with 95% confidence intervals.
Etiologies of extensor mechanism failure and indications for RTKA.
Surgical technique
The entire surgery is available on Video 1.
Preoperative planning
frozen extensor mechanism allografts were preferred for their superior biological properties, and fresh-frozen grafts were favored over irradiated ones [8]. The graft dimensions were appropriate to restore patellar thickness, width, and height, as well as the anatomical dimensions of the tendons, which were evaluated using radiographs of the contralateral knee. Particular attention was paid to the dimensions of the tibial tubercle osteotomy (TTO) and the length of the quadriceps tendon (QT). After thawing and before skin incision, the graft was inspected, measured, and a systematic bacteriological sample was taken.
Surgical approach
The patient was positioned supine, with the operative leg draped in the field, supported by a foot support and a lateral support, without a tourniquet. In patients with multiple prior incisions, the most lateral one was reopened. Arthrotomy was performed through the area of the extensor mechanism rupture. A patellectomy was carried out. If the native patellar tendon was still present, it was split longitudinally up to the anterior tibial tuberosity. The native patellar retinaculum and prepatellar fascia were preserved to enhance graft coverage. The previous TKA components were carefully removed using saws and chisels to minimize bone loss. Residual cement and any intramedullary plugs were also removed.
Allograft preparation
The TTO of the allograft was shaped into a bone block of approximately 6 × 1.5 × 2 cm [9] (Figure 1). The QT was split into two bundles, using traction sutures [10].
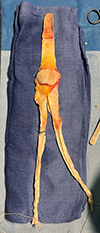 |
Figure 1 Allograft preparation. |
Reimplantation
The first step following removal of the previous TKA components was evaluation of bone loss prior to new femoral and tibial bone cuts [11]. The second step was then the revision of bone cuts. The third step was the choice of implants and level of constraint, tailored to each case. In situations involving moderate to severe bone loss, metaphyseal cones were used to enhance fixation. The use of metallic wedges was based on the extent and location of bone deficiency (Figure 2). The fourth step consisted of trials positioning and coronal laxity in flexion and extension was tested.
Trials in place, the fifth step was preparation of the future TTO site, using saws and chisels to create a receiving socket matching the size of the allograft. The target dimensions were approximately 6 cm in length, 1.5 cm in depth, and 2 cm in width, with clean edges to allow press-fit fixation [9] (Video 2). Then trials were removed, and the sixth step consisted of cementation. All implants, except for cones, were cemented using antibiotic-loaded cement and diaphyseal plugs. Particular attention was paid to tibial osteotomy to prevent cement leakage.
 |
Figure 2 Example of RTKA: semi-constrained prosthesis with distal wedges, stems, and tibial cone (A, B), tibial and femoral cones in situ (C). |
Allograft
Fixation of the allograft was performed with the knee in full extension. The graft was initially impacted for a press-fit and then fixed to the tibia using two to three screws (3.5 or 4.5 mm in diameter) [9] (Video 3). Metal washers might be added if necessary. For proximal fixation, the two bundles of the QT graft were passed through the native QT using three weaves per limb, according to the Pulvertaft technique. The graft was then secured with multiple non-absorbable sutures. During suturing, tension was applied in opposite directions on the allograft and native QT strands to ensure optimal tensioning of the reconstructed extensor mechanism (Figure 3). The native patellar retinacula, the prepatellar fascia, and, if present, the native patellar tendon were then sutured to the allograft. (Video 4). The patella of the allograft was not resurfaced to avoid the risk of graft osteolysis. Patellar tracking was assessed in slight flexion to prevent displacement of fixation screws or fracture of the tibial allograft bone block before graft sutures. Intraoperative fluoroscopy was used to check patellar height and screws positioning.
 |
Figure 3 TTO fixation (A, B, C). A: anterior tibial tuberosity fixation. (B), (C): proximal fixation: the two bundles of the QT graft are passed through the native QT using three weaves per limb, according to the Pulvertaft technique. The graft was then secured with multiple non-absorbable sutures. During suturing, tension was applied in opposite directions on the allograft and native QT strands to ensure optimal tensioning of the reconstructed extensor mechanism. |
Postoperative care
Following radiological and clinical assessment, a knee brace in full extension was required for 3 months (Figure 4). Weight-bearing was permitted with the use of crutches and brace. Knee flexion was permitted in a gradual manner starting from the third postoperative month with 30-degree increments every 2 weeks. A hinged brace was maintained until full quadriceps locking was achieved. The rehabilitation protocol was supervised by a specialised physiotherapist for a minimum of six months to one year.
The minimum postoperative follow-up included radiological and clinical evaluations at 2, 3, and 6 months, with subsequent evaluations at 1 and 2 years post-surgery (Video 5).
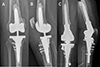 |
Figure 4 Postoperative radiographs: example of two different types of constraint. A, B: Varus Valgus Constraint implant; C, D: Rotating hinge. In both cases, TTO fixation was possible using screws despite tibial cone or sleeve. |
Results
Our cohort consisted of 20 patients, including 11 women (55%) and 9 men (45%), with a mean age of 65.3 years. As major risk factors, two patients (10%) were smokers, five patients (25%) had a BMI ≥ 30. The average follow-up duration was 31.6 months.
The cohort included 19 RTKA procedures (95%) and one revision of a femoro-patellar prosthesis (PFP) to a TKA (5%). Various implant types were used: 1 posterior-stabilized prosthesis (5%), 2 semi-constrained prostheses (10%), 15 rotating-hinge prostheses (75%), and 2 resection prostheses (10%).
Complications and etiologies of failure are listed in Table 2. Regarding postoperative complications, seven patients (35%) developed a total of eight complications related to RTKA. Five patients (25%) experienced rupture of the extensor mechanism allograft at different levels. Nine patients (45%) experienced septic complications. Fifteen patients (75%) underwent at least one additional surgical procedure due to complications. Functional flexion (>90°) was generally achieved between the fourth and sixth postoperative months. Mean flexion at last follow-up was 109° (70–130°).
The survival rate without failure was 60% at 12 months. The mean survival time without failure was 18.35 months. The survival rate without allograft failure was 78.6% at 12 months. Infection-free survival rate (excluding DAIR cases) was 72.7% at 12 months and remained stable at the final follow-up.
Results, complications, and failures.
Discussion
The high complication and failure rates reflect the complexity of this highly selected cohort of multiply operated knees. In this setting, the procedure should not be regarded as a routine indication but rather as a salvage strategy prior to arthrodesis, definitive implant removal, or transfemoral amputation [12]. The goal is not to achieve optimal function, but to restore ambulation, active extension and flexion, and to reduce pain and disability.
No study has specifically evaluated total extensor mechanism allograft reconstruction combined with single-stage RTKA. Browne and Hanssen [13] reported a 38% failure rate in 50 reconstructions, with 56% 10-year failure-free survival. Complications included 8% graft rupture, 10% infection, and 20% extensor lag ≥30°. Ricciardi et al. [7] observed a 58% reoperation rate at 68 months in 26 knees, 19% of which were associated with RTKA.
Synthetic grafts appear to provide comparable results. Shau et al. [14], in a meta-analysis of 204 knees, found similar success rates for allografts (76%) and synthetic grafts (74%), without differences in complications. Synthetic grafts were more frequently used when revision TKA was required, likely because they allow simpler distal fixation. Achilles tendon allografts have shown comparable outcomes, with a 58.6% success rate at 42 months in 29 knees [15]. We nevertheless favor total extensor mechanism allografts for their more reliable proximal fixation and improved native tissue coverage.
All reconstruction techniques are associated with high rates of infection. The combination of RTKA and extensor mechanism reconstruction likely increases this risk due to prolonged operative time and soft tissue compromise.
This is, to our knowledge, the first study specifically addressing total extensor mechanism allografts combined with RTKA. Its limitations include a small sample size and a retrospective design, and the fact that subgroup analysis was not feasible.
Conclusion
Total extensor mechanism allograft reconstruction combined with single-stage revision TKA is a demanding salvage procedure that requires meticulous stepwise surgical execution. Key technical aspects include accurate graft sizing, secure tibial tubercle fixation with press-fit and screw stabilization, precise proximal fixation using the Pulvertaft technique under appropriate tension, preservation of native soft tissue coverage, and strict postoperative immobilization.
When these technical principles are respected, this procedure provides a limb-preserving alternative to arthrodesis in complex, multiply operated knees. Despite the high complication rate inherent to this challenging population, it remains a valuable option in specialized referral centers, provided that patients are carefully selected and adequately informed of the surgery's salvage nature.
Funding
This research did not receive any specific funding.
Conflicts of interest
Author 1 and Author 2 declare that they have no relevant financial or non-financial interests to report. Author 3: Consultancy from Smith and Nephew, Groupe Lepine and Stryker. Author 4: Consultancy from Smith and Nephew. Author 5: Consultancy from Stryker, Heraeus: Institutional research support from Groupe Lepine, Amplitude; Editorial Board for SICOTJ and MO Journal.
Data availability statement
Data generated and/or analyzed during this study are not publicly available due to legal and ethical restrictions related to patient confidentiality. All photographs and videos related to the surgical technique are available within the article and its supplementary material.
Author contribution statement
Author 1: conceptualization, methodology, investigation, writing, editing.
Author 2: conceptualization, methodology, writing.
Author 3: conceptualization, methodology, reviewing, editing.
Author 4: conceptualization, reviewing.
Author 5: conceptualization, reviewing, editing.
Ethics approval
All procedures were performed in accordance with the ethical standards of the institutional and/or national research committee, the 1964 Helsinki declaration and its later amendments, or comparable ethical standards. Data collection and analysis were carried out in accordance with MR004 Reference Methodology from the CNIL (Ref. 2238218v0) obtained on March 24, 2025. The study was registered and filed on the Health Data Hub website.
Informed consent
Written informed consent was obtained from all patients.
Supplementary material
Video 1. Complete case surgery.
Video 2. Preparation of tibial socket for allograft.
Video 3. Knee testing after fixation of the allograft.
Video 4. Native apparal suture on allograft in tension.
Video 5. Walking outcome at six months.
Access Supplementary MaterialReferences
- Schoderbek RJ, Brown TE, Mulhall KJ, et al. (2006) Extensor mechanism disruption after total knee arthroplasty. Clin Orthop Relat Res 446, 176–185. [Google Scholar]
- Bonnin M, Chouteau J, Mouttet C, et al. (2016) Extensor tendon ruptures after total knee arthroplasty. Orthop Traumatol Surg Res 102(3), 313–318. [Google Scholar]
- Fiquet C, Schneider A, Ballis R, et al. (2017) Reconstructing the chronically disrupted knee extensor mechanism after total knee arthroplasty: hourglass variant of the original partial allograft technique. Orthop Traumatol Surg Res 103(6), 791–797. [Google Scholar]
- Brown NM, Murray T, Sporer SM, et al. (2015) Extensor mechanism allograft reconstruction for extensor mechanism failure following total knee arthroplasty. J Bone Joint Surg Am 97(4), 279–283. [Google Scholar]
- Sain A, Bansal H, Pattabiraman K, et al. (2021) Extensor mechanism reconstruction using allograft following total knee arthroplasty: a review of current practice. Cureus 13(1), e12803. [Google Scholar]
- Emerson RH Jr, Head WC, Malinin TI (1990) Reconstruction of patellar tendon rupture after total knee arthroplasty with an extensor mechanism allograft. Clin Orthop Relat Res 254, 154–161. [Google Scholar]
- Ricciardi BF, Oi K, Trivellas M, et al. (2016) Survivorship and prognostic factors for extensor mechanism allograft reconstruction after total knee replacement. J Arthroplasty 31(9), 2039–2044. [Google Scholar]
- Rihn JA, Harner CD (2003) The use of musculoskeletal allograft tissue in knee surgery. Arthroscopy 19(Suppl 1), 51–66. [Google Scholar]
- Cance N, Batailler C, Canetti R, et al. (2023) Tibial tubercle osteotomy during the revision of total knee arthroplasty: the technique of a referral center with 10 years of experience. SICOT J 9, 18. [Google Scholar]
- Murgier J, Boisrenoult P, Pujol N, et al. (2015) Knee extensor mechanism allograft reconstruction following chronic disruption, Orthop Traumatol Surg Res. 101(7), 867–870. [Google Scholar]
- Engh GA, Ammeen DJ (1999) Bone loss with revision total knee arthroplasty: defect classification and alternatives for reconstruction. Instr Course Lect, 48, 167–175. [Google Scholar]
- Haidukewych GJ, Petrie JR, Adigweme O (2014) The multiply-operated total knee replacement patient : salvage options. Bone Joint J 96-B(Suppl A), 122–124. [Google Scholar]
- Browne JA, Hanssen AD (2011) Reconstruction of patellar tendon disruption after total knee arthroplasty : results of a new technique utilizing synthetic mesh, J Bone Joint Surg Am 93(12), 1137–1143. [Google Scholar]
- Shau D, Patton R, Patel S et al. (2018) Synthetic mesh vs. allograft extensor mechanism reconstruction in total knee arthroplasty - A systematic review of the literature and meta-analysis. Knee 25(1), 2–7. [Google Scholar]
- Diaz-Ledezma C, Orozco FR, Delasotta LA et al. (2014) Extensor mechanism reconstruction with achilles tendon allograft in TKA results of an abbreviate rehabilitation protocol. J Arthroplasty 29(6), 1211–1215. [Google Scholar]
Cite this article as: Salese Y, Delfosse G, Batailler C, Servien E & Lustig S (2026) Single-stage total knee arthroplasty revision with extensor mechanism allograft: surgical technique. SICOT-J 12, 26. https://doi.org/10.1051/sicotj/2026014.
All Tables
All Figures
 |
Figure 1 Allograft preparation. |
| In the text | |
 |
Figure 2 Example of RTKA: semi-constrained prosthesis with distal wedges, stems, and tibial cone (A, B), tibial and femoral cones in situ (C). |
| In the text | |
 |
Figure 3 TTO fixation (A, B, C). A: anterior tibial tuberosity fixation. (B), (C): proximal fixation: the two bundles of the QT graft are passed through the native QT using three weaves per limb, according to the Pulvertaft technique. The graft was then secured with multiple non-absorbable sutures. During suturing, tension was applied in opposite directions on the allograft and native QT strands to ensure optimal tensioning of the reconstructed extensor mechanism. |
| In the text | |
 |
Figure 4 Postoperative radiographs: example of two different types of constraint. A, B: Varus Valgus Constraint implant; C, D: Rotating hinge. In both cases, TTO fixation was possible using screws despite tibial cone or sleeve. |
| In the text | |
Current usage metrics show cumulative count of Article Views (full-text article views including HTML views, PDF and ePub downloads, according to the available data) and Abstracts Views on Vision4Press platform.
Data correspond to usage on the plateform after 2015. The current usage metrics is available 48-96 hours after online publication and is updated daily on week days.
Initial download of the metrics may take a while.


